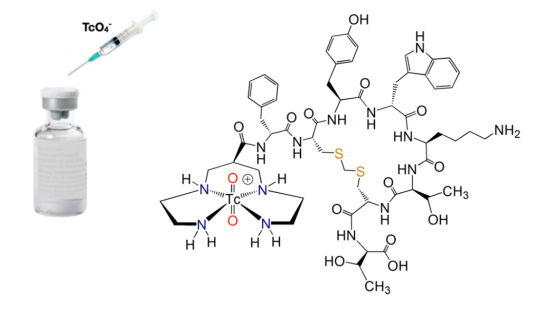
Despite advances in radiopharmaceuticals for diagnosis and therapy, technetium-99m (99mTc) radiopharmaceuticals are still the major diagnostic tools in clinical nuclear medicine worldwide. They are one of the most important nuclear applications in the field of human health, playing a crucial role in the diagnosis of cancer, cardiac and various neurological disorders. Molybdenum-99/Technetium-99m (99Mo/99mTc) generators are the major source of 99mTc for radiopharmaceutical production. They are usually derived from Mo-99 through uranium fission reaction. It is likely that 99mTc radioisotopes, produced via many routes, will continue to be the major source of radiopharmaceutical production in the coming decades.
Development of new targeting molecules such as prostate-specific membrane antigen (PSMA) ligands for the detection and treatment of prostate cancers; fibroblast activation protein inhibitors (FAPI), highly expressed in the major cell population in tumours; as well as somatostatin ligands for human neuroendocrinal tumours, have revolutionized the oncology practice worldwide. Special considerations should be taken to disseminate, translate and develop production and application of related 99mTc analogues. Growing challenges to human health from neurological diseases (e.g., Parkinson’s, Alzheimer’s) and infectious diseases (viral, fungal and bacterial) could benefit from the development of new tracers, especially tracers based on widely available 99mTc radiopharmaceuticals. Making 99mTc radiopharmaceutical facilities and SPECT imaging centres available worldwide, as well as new advances in imaging techniques, could enable many countries to benefit from these crucial tools for nuclear medicine.
This IAEA Coordinated Research Project (CRP) will establish a global network of producers and researchers for developing a new generation of 99mTc kits and radiopharmaceuticals. The CRP is expected to develop standard procedures and guidelines for the production, quality control and preclinical studies of the novel 99mTc radiopharmaceuticals. The CRP will also focus on the most clinically relevant items on demand and application of new radiolabelling strategies.
CRP Overall Objective
The CRP will focus on transferring the knowledge acquired to produce a series of 99mTc radiopharmaceuticals for imaging various biological substrates of relevant clinical interest using the most efficient methods of 99mTc labelling. Several 99mTc radiopharmaceuticals will be produced for imaging the following biological substrates.
Specific Research Objectives
- Research and development on 99mTc PSMA receptors ligands
- Research and development on 99mTc based peptides or small molecules targeting receptors expressed by cancerous lesions classified as poor-prognostic solid tumours such as breast cancer
- Tumour microenvironment studies using newly developed ligands
- Round robins comprised of labelling kit formulations previously developed for the preparation of 99mTc radiopharmaceuticals used in myocardial perfusion, bone imaging, tumour imaging, infection processes and sentinel lymph node detection
- Identification of suitable and efficient quality control procedures and pre-clinical assays for assessing the overall quality of 99mTc radiopharmaceuticals prepared during the CRP
- Enhancement of capacity building for applying freeze-drying methods and technologies for the development of lyophilized kit formulations of 99mTc radiopharmaceuticals investigated during the CRP
- Non-clinical studies including in vitro cell studies and in vivo animal studies for evaluating stability, dosimetry, therapeutic efficacy, and possible side effects.
- Preparation and formulation of appropriate protocols and guidelines accessible for all IAEA Member States
Criteria
- Availability of 99mTc generators
- Labelling and quality control infrastructure
- Previous experience in labelling and quality control of 99mTc
- Availability of pre-clinical facilities
- Availability of freeze-drying equipment
How to join this CRP
Interested research centres must submit their Proposal for Research Contract or Agreement by email to the IAEA’s Research Contracts Administration Section, using the appropriate template on the CRA website no later than 31 March 2024. For further information related to this CRP, potential applicants should use the contact form under the CRP page.