
If you would like to learn more about the IAEA’s work, sign up for our weekly updates containing our most important news, multimedia and more.
Neutrons for Neurons and Cyclotrons for Radioisotopes
Michael Madsen
Glioblastoma is an aggressive malignancy and accounts for about 15 per cent of all brain tumours. Even when initially controlled by treatment, the cancer almost always returns. Surgery and radiotherapy can extend survival by some months, but the brain cancer usually ends life within one to two years of diagnosis, and less than five per cent of people survive longer than five years. As with glioblastoma, many cranial cancers, are a challenge to treat due to the sensitive nature of normal brain tissue to surgery and radiotherapy, but there is hope that could change soon, thanks in part to new therapies made possible by accelerators producing intense sources of neutrons.
“When you think of performing nuclear reactions, you probably don’t imagine a human head is the best place to do it — but you’d be wrong,” said Ian Swainson, a nuclear physicist at the IAEA. He is helping develop IAEA guidance on accelerator applications for neutron production, including in medicine. He said using this technology in one cancer therapy in particular, boron neutron capture therapy (BNCT), is very promising: “Firing neutrons at boron atoms in certain brain, head and neck cancers can save lives.”
“ Firing neutrons at boron atoms in certain brain, head and neck cancers can save lives.
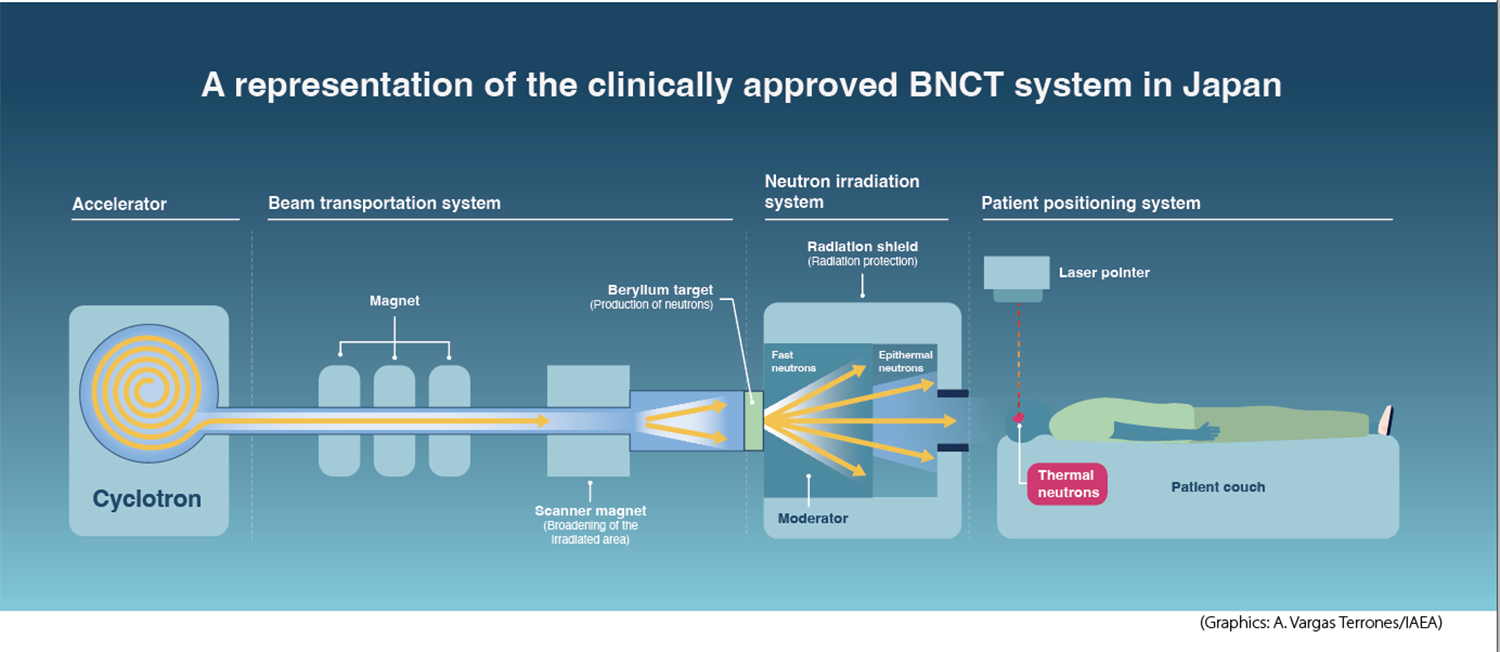
BNCT uses the destructive power that neutrons can unleash and relies on localizing damage to the tumour as much as possible. Harnessing neutrons’ destructive ability is possible with boron-10 isotopes. “Boron-10 is non-radioactive and great at capturing neutrons. As a result, in a very localized nuclear reaction, boron breaks into two energetic fragments. So, by injecting a patient with special drugs that deliver boron-10 to tumour sites, we’re putting a big bullseye on the cancer,” Swainson explained.
Still largely experimental, BNCT is not widely available, but that is changing. In 2020, two BNCT facilities began clinical treatments in Koriyama and Osaka in Japan. In the same year, the IAEA and Japan’s Okayama University agreed to enhance cooperation in BNCT through events, knowledge and information exchange, and the development of a BNCT facilities database.
“BNCT is a cutting-edge cancer therapy,” Hirofumi Makino, President of Okayama University, said at the time. “It is a happy marriage of modern nuclear physics and up-to-date pharmaceutical cell biology. However, we should not forget the long history of struggle in developing this difficult medical technology.”
In 2001, the IAEA produced a technical report on BNCT, which became a reference work for the field. At that time, the only neutron sources involved were research reactors. Since then, a new generation of compact accelerator-based neutron sources has been developed, which can be installed directly in clinics. This has led to a major resurgence in interest in BNCT.
BNCT projects are also being pursued in Argentina, China, Finland and the Republic of Korea. “20 years ago, using neutrons from accelerators in cancer treatment was just theory. Now it’s reality and we’re reflecting this development in an upcoming technical document entitled Advances in Boron Neutron Capture Therapy,” said Swainson.
The cyclotron revolution
Determining the feasibility of BNCT in a patient requires injecting a boron compound radiolabelled with fluorine-18 (18F), produced with cyclotrons, followed by imaging the patient using a nuclear medicine technique called positron emission tomography–computed tomography (PET–CT). The 18F labelled compound is called 4-borono-2-18F-fluoro-phenylalanine, or FBPA.
“FBPA is important because it confirms to doctors that a tumour has absorbed a boron containing compound and is ready for BNCT. Without it, the therapy may not work. As BNCT becomes more widely available, we’re going to need cyclotrons to meet FBPA demand,” said Amirreza Jalilian, a radioisotope and radiopharmaceutical chemist at the IAEA. A cyclotron is a type of particle accelerator that produces radioisotopes used in nuclear medicine by firing a particle beam at stable isotopes. The interaction results in a nuclear reaction that creates short-lived radioisotopes. Because these radioisotopes decay quickly, they need to be produced near or on the site where treatment takes place and used right away.
Jalilian notes that, although the number of research reactors used in the production of radioisotopes is rather stable, new, versatile and increasingly affordable cyclotrons are on the rise worldwide. Many of the short-lived radioisotopes used in patients can be produced by cyclotrons in hospitals, and that is a major advantage for the technology.
The radiopharmaceutical fluorodeoxyglucose is just one example. It relies on 18F, which can be produced with cyclotrons. That radiotracer is used in around 95 per cent of PET–CT procedures, and so is critical in neuroimaging and diagnosing cancer.
Another workhorse among radiopharmaceuticals is gallium-68 (68Ga), which is the key component of some theranostic radiopharmaceuticals — a type of pharmaceutical that uses radioisotopes both for diagnosis and therapy through the release of radiation. Such radiopharmaceuticals play an important role in diagnosing and following up on cancers, and shows particular promise in addressing prostate cancer. Producing 68Ga, however, does have its challenges.
“Today, the most common method for producing 68Ga is with a non-accelerator system called a generator, but generators simply cannot produce enough to meet demand. Cyclotrons offer an effective alternative means of direct production and are already greatly expanding the availability of 68Ga,” said Jalilian, explaining that ten centres around the world are now routinely using cyclotrons to produce 68Ga. The IAEA is currently coordinating a research project to support the exchange of international expertise in cyclotron-based production of 68Ga, and in 2019 released Gallium-68 Cyclotron Production, a publication dedicated to the topic.






